Use of blockchain in the production of medicinal cannabis
This article – published in TechnoPharm 11, Nr. 1, 216–221 (2021), Editio Cantor Verlag, Aulendorf (Germany) – describes how the production of medicinal cannabis can be optimised through the fully integrated use of blockchain technology in the manufacturing and transport processes, thus creating a continuous data and information network. It shows how blockchain can be applied in the regulatory challenging production of medicinal cannabis, increasing traceability and quality of the products. By applying digital technology along the entire value chain, the identity of the medicinal product can be proven clearly and counterfeit-proof, and patient delivery can be made more secure.
by Rainer Krüger and Dr. Lisann Eßer, J&K Consulting GmbH, Rommerskirchen (Germany)
Secure data management in the pharmaceutical industry
During pharmaceutical production, large amounts of data are generated, such as process or product-relevant data. The management of the ever-in- creasing amount of data is a major challenge for the pharmaceutical industry, as the demand for data integrity, security, and clarity, specified by guidelines and laws, is very high. Within the documentation systems of pharmaceutical companies, guaran- teeing data integrity and clarity is often only possible with great effort. Difficulties arise especially when data are processed by several people and passed on via different departments or locations. In addition, the secure and integrity exchange of data when collaborating with external organisations, such as suppliers, is often a major challenge due to different software applications.
Currently, most data are stored in a central system. This means that most of the data is stored on a central server, which users can access from any location with the appropriate access data. Whereas only a small amount of data is stored on the individual user devices. Well-known examples of central data storage are cloud or messenger services. The problem here is the security of the data and protection against unauthorised access, because where large amounts of data are stored, there is a risk of data misuse.
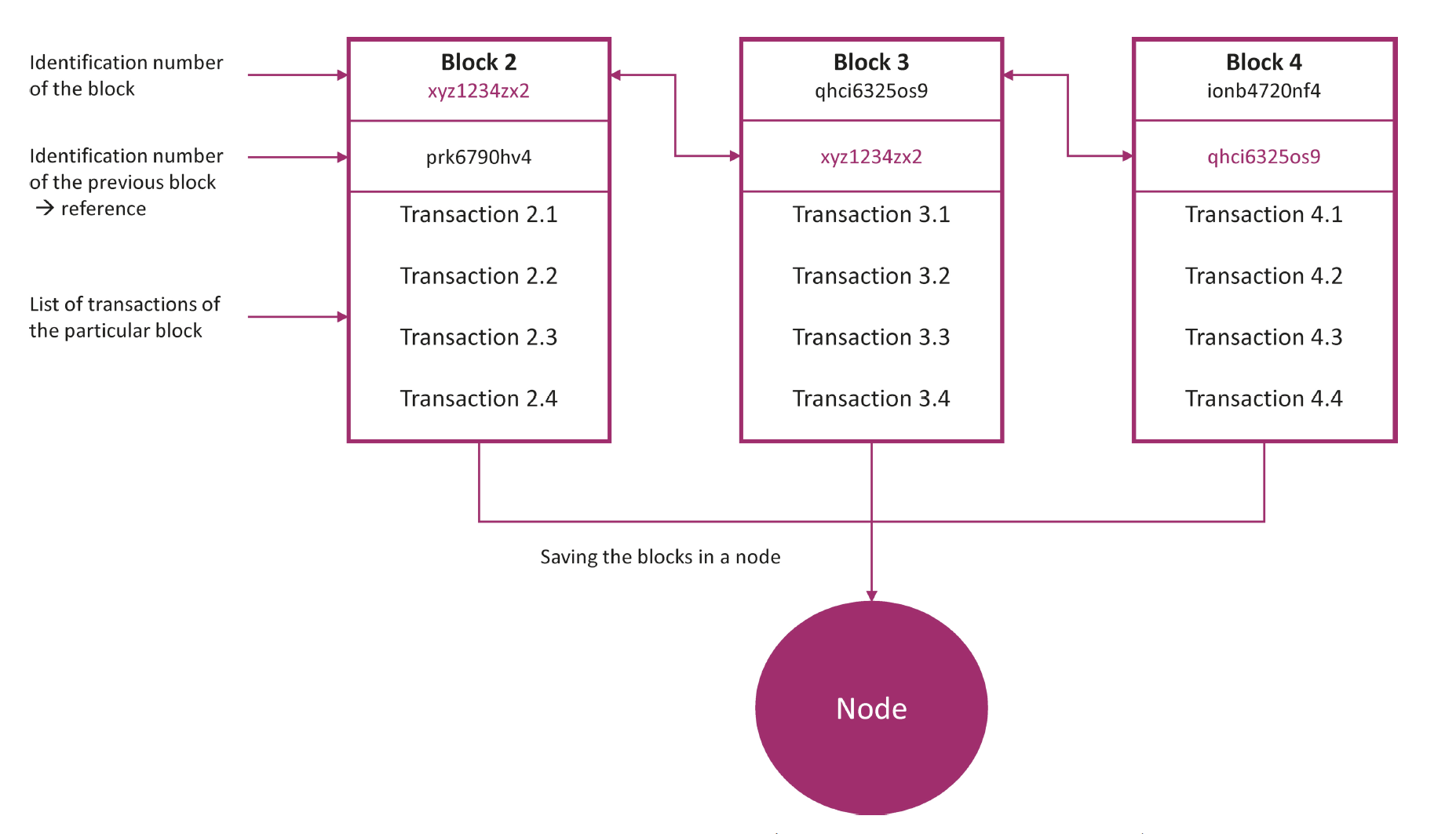
One solution to the problems described above is the decentralised storage of data in a blockchain. In the blockchain, data records, known as blocks, are stored decentrally, i.e., locally on the end devices of the users. As soon as a block is generated, the block is verified and stored by the computers in the network. The computers represent the (network) nodes, i.e., the connection points for data transmission. The blocks are chrono- logically and linearly expandable and are chained together using crypto- graphic procedures. The verified block is cryptographically encrypted by an algorithm and attached to a chain of data records (blockchain). Since the blocks also have a digital fingerprint that has a timestamp and the transac- tion data, many unique data records are created with their own traceable history ( fig. 1). Therefore, the block- chain is a secure and always up-to- date register in which digital actions can be documented reliably and clearly for all participants.
With the help of the blockchain, product parameters, facility characteristics or process data, for example, can be shared and jointly processed in the pharmaceutical industry with a desired group of people securely and while guaranteeing data integrity. If the data records are updated with new process data, for example, these can be verified and saved with a time stamp. If the product then changes owners because it is transported further along the supply chain, this transaction is also documented. This opens possibilities for the user to document processes and supply chains securely and without gaps, and especially in the special case of medicinal cannabis.
>>> Download the entire article here (by courtesy of the publisher)
Published: TechnoPharm 11, Nr. 1, 216–221 (202
Photo Credit: J&K Consulting GmbH